(Whole Blood /Serum/Plasma) Package Insert |
| For professional in vitro diagnostic use only. A rapid test for the diagnosis of Syphilis to detect antibodies (IgG and IgM) to Treponema Pallidum (TP) qualitatively in whole blood.Serum or plasma. |
| [USO PREVISTO] The Syphilis Rapid Test Cassette (Whole Blood/Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of antibodies (IgG and IgM) to Treponema Pallidum (TP) in whole blood, serum or plasma to aid in the diagnosis of Syphilis. |
| [SUMMARY] Treponema Pallidum (TP) is the causative agent of the venereal disease Syphilis. TP is a spirochete bacterium with an outer envelope and a cytoplasmic membrane.1 Relatively little is known about the organism in comparison with other bacterial pathogens. According to the Center for Disease Control (CDC), the number of cases of Syphilis infection has markedly increased since 1985.2 Some key factors that have contributed to this rise include the crack cocaine epidemic and the high incidence of prostitution among drug users.3 One study reported a substantial epidemiological correlation between the acquisition and transmission of the HIV virus and Syphilis.4 Multiple clinical stages and long periods of latent, asymptomatic infection are characteristic of Syphilis. Primary Syphilis is defined by the presence of a chancre at the site of inoculation. The antibodies response to the TP bacterium can be detected within 4 to 7 days after the chancre appears. The infection remains detectable until the patient receives adequate treatment.5 The Syphilis Rapid Test Cassette (Whole Blood/Serum/Plasma) utilizes a double antigen combination of a Syphilis antigen coated particle and Syphilis antigen immobilized on membrane to detect TP antibodies (IgG and IgM) qualitatively and selectively in whole blood, serum or plasma. |
| [PRINCIPLE] The Syphilis Rapid Test Cassette (Whole Blood/Serum/Plasma) is a qualitative membrane based immunoassay for the detection of TP antibodies (IgG and IgM) in whole blood, serum or plasma. In this test procedure, recombinant Syphilis antigen is immobilized in the test line region of the test. After specimen is added to the specimen well of the cassette, it reacts with Syphilis antigen coated particles in the test. This mixture migrates chromatographically along the length of the test and interacts with the immobilized Syphilis antigen. The double antigen test format can detect both IgG and IgM in specimens. If the specimen contains TP antibodies, a colored line will appear in the test line region, indicating a positive result. If the specimen does not contain TP antibodies, a colored line will not appear in this region, indicating a negative result. To serve as a procedural control, a colored line will always appear in the control line region, indicating that proper volume of specimen has been added and membrane wicking has occurred. |
| [REAGENTS] The test contains Syphilis antigen coated particles and Syphilis antigen coated on the membrane. |
| [PRECAUTIONS] | |
| * | For professional in vitro diagnostic use only. Do not use after expiration date. |
| * | Do not eat, drink or smoke in the area where the specimens or kits are handled. |
| * | Do not use test if pouch is damaged. |
| * | Handle all specimens as if they contain infectious agents. Observe established precautions against microbiological hazards throughout all procedures and follow the standard procedures for proper disposal of specimens. |
| * | Wear protective clothing such as laboratory coats, disposable gloves and eye protection when specimens are assayed. |
| * | The used test should be discarded according to local regulations. |
| * | Humidity and temperature can adversely affect results. |
| [STORAGE AND STABILITY] | |
| Store as packaged in the sealed pouch either at room temperature or
refrigerated (2-30°C). The test is stable through the expiration date printed on the sealed pouch. The test must remain in the
sealed pouch until use. DO NOT FREEZE. Do not use after the expiration date. |
| [SPECIMEN COLLECTION AND PREPARATION] | |||
| * | The Syphilis Rapid Test Cassette (Whole Blood/Serum/Plasma) can be performed using whole blood (from venipuncture or fingerstick), serum or plasma. | ||
| * | To collect Fingerstick Whole Blood specimens: | ||
| • | Wash the patient's hand with soap and warm water or clean with an alcohol swab. Allow to dry. | ||
| • | Massage the hand without touching the puncture site by rubbing down the hand towards the fingertip of the middle or ring finger. | ||
| • | Puncture the skin with a sterile lancet. Wipe away the first sign of blood. | ||
| • | Gently rub the hand from wrist to palm to finger to form a rounded drop of blood over the puncture site. | ||
| • | Add the Fingerstick Whole Blood specimen to the test by using a capillary tube: | ||
| ⚬ | Touch the end of the capillary tube to the blood until filled to approximately 80µL. Avoid air bubbles. | ||
| ⚬ | Place the bulb onto the top end of the capillary tube, then squeeze the bulb to dispense the whole blood to the specimen area of the test cassette. | ||
| • | Add the Fingerstick Whole Blood specimen to the test by using hanging drops: | ||
| ⚬ | Position the patient's finger so that the drop of blood is just above the specimen area of the test cassette. | ||
| ⚬ | Allow 2 hanging drops of fingerstick whole blood to fall into the center of the specimen area on the test cassette, or move the patient's finger so that the hanging drop touches the center of the specimen area. Avoid touching the finger directly to the specimen area. | ||
| * | Separate serum or plasma from blood as soon as possible to avoid hemolysis. Use only clear non-hemolyzed specimens. | ||
| * | Testing should be performed immediately after the specimens have been collected. Do not leave the specimens at room temperature for prolonged periods. Serum and plasma specimens may be stored at 2-8°C for up to 3 days. For long term storage, specimens should be kept below -20°C. Whole blood collected by venipuncture should be stored at 2-8°C if the test is to be run within 2 days of collection. Do not freeze whole blood specimens. Whole blood collected by fingerstick should be tested immediately. | ||
| * | Bring specimens to room temperature prior to testing. Frozen specimens must be completely thawed and mixed well prior to testing. Specimens should not be frozen and thawed repeatedly. | ||
| * | If specimens are to be shipped, they should be packed in compliance with local regulations covering the transportation of etiologic agents. | ||
| [MATERIALS] | |
| Materials provided | |
| • Test Cassettes • Droppers • Buffer • Package insert |
|
| Materials required but not provided | |
| • Specimen collection Containers • Centrifuge • Timer • Lancets • Heparinized capillary tubes and dispensing bulb |
| [DIRECTIONS FOR USE] |
|
| Allow the test, specimen, buffer and/or controls to reach room temperature (15-30°C) prior to testing. | |
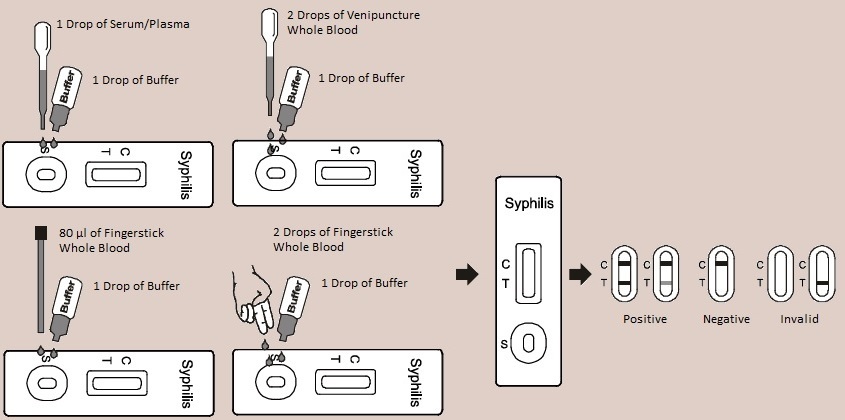
| 1. | Bring the pouch to room temperature before opening it. Remove the test cassette from the sealed pouch and use it as soon as possible. |
| 2. | Place the cassette on a clean and level surface. For Serum or Plasma specimen: Hold the dropper vertically and transfer 1 drop of serum or plasma (approximately 40 µL) to the specimen area, then add 1 drop of buffer (approximately 40 µL),and start the timer, see illustration below. For Venipuncture Whole Blood specimen: Hold the dropper vertically and transfer 2 drops of whole blood (approximately 80 µL) to the specimen area, then add 1 drop of buffer (approximately 40 µL), and start the timer. See illustration below. For Fingerstick Whole Blood specimen: |
| • | To use a capillary tube: Fill the capillary tube and transfer approximately 80 µL of fingerstick whole blood specimen to the specimen area of test cassette, then add 1 drop of buffer (approximately 40 µL) and start the timer. See illustration below. | ||
| • | To use hanging drops: Allow 2 hanging drops of fingerstick whole blood specimen (approximately 80 µL) to fall into the specimen area of test cassette, then add 1 drop of buffer (approximately 40 µL) and start the timer. See illustration below. | ||
| 3. | Wait for the colored line(s) to appear. Read results at 5 minutes.
Do not interpret the result after 20 minutes. |
 |
|
| [INTERPRETATION OF RESULTS] |
|
| POSITIVE:* Two lines appear. One colored line should be
in the control line region (C) and another apparent colored line should be in the test line region (T). *NOTE: The intensity of the color in the test line region (T) will vary depending on the concentration of TP antibodies present in the specimen. Therefore, any shade of color in the test line region (T) should be considered positive. NEGATIVE: One colored line appears in the control line region (C). No line appears in the test line region (T). INVALID: Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the procedure and repeat the test with a new test. If the problem persists, discontinue using the test kit immediately and contact your local distributor. |
|
| [QUALITY CONTROL] A procedural control is included in the test. A colored line appearing in the control line region (C) is considered an internal procedural control. It confirms sufficient specimen volume, adequate membrane wicking and correct procedural technique. |
|
| Control standards are not supplied with this kit; however, it is recommended
that positive and negative controls be tested as a good laboratory practice to confirm the test procedure and to verify proper test performance. |
|
| [LIMITATIONS] |
|
| 1. | The Syphilis Rapid Test Cassette (Whole Blood/Serum/Plasma) is for in vitro diagnostic use only. The test should be used for the detection of TP antibodies in whole blood, serum or plasma specimens only. Neither the quantitative value nor the rate of increase in TP antibodies can be determined by this qualitative test. |
| 2. | The Syphilis Rapid Test Cassette (Whole Blood/Serum/Plasma) will only indicate the presence of TP antibodies in the specimen and should not be used as the sole criteria for the diagnosis of TP infection. |
| 3. | As with all diagnostic tests, all results must be interpreted together with other clinical information available to the physician. |
| 4. | If the test result is negative and clinical symptoms persist, additional
testing using other clinical methods is recommended. A negative result does not at any time preclude the possibility of TP infection. |
| [EXPECTED VALUES] | |
| The Syphilis Rapid Test Cassette (Whole Blood/Serum/Plasma) has been compared
with a leading commercial TPPA Syphilis test, demonstrating an overall accuracy greater than or equal to 99.8%. |
|
[PERFORMANCE CHARACTERISTICS]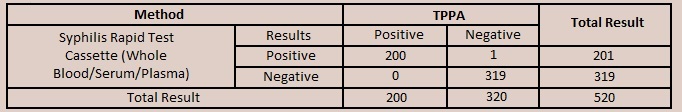 |
| Relative Sensitivity: >99.9% (95%CI*: 99.4%-100%) Relative Specificity: 99.7% (95%CI*: 98.3%-100%) Accuracy: 99.8% (95%CI*: 98.9%-100%) |
*Confidence Interval |
Intra-Assay |
|
| Acetaminophen: 20 mg/dL Acetylsalicylic Acid: 20 mg/dL Ascorbic Acid: 2g/dL Creatin: 200 mg/dL Bilirubin: 1g/dL |
Caffeine: 20 mg/dL Gentisic Acid: 20 mg/dL Albumin: 2 g/dL Hemoglobin 1.1 mg/dL Oxalic Acid: 600mg/dL |
| None of the substances at the concentration tested interfered
in the assay. |
|
| [BIBLIOGRAPHY] | |
| 1. | Claire M. Fraser. Complete genome sequence of Treponema Pallidum, the Syphilis spirochete, Science 1998; 281 July: 375-381 |
| 2. | Center for Disease Control. Recommendations for diagnosing and treating Syphilis in HIV infected patients, MMWR Morb. Mortal Wkly Rep. 1988; 37: 601 |
| 3. | Aral R. Marx. Crack, sex and STD, Sexually Transmitted Diseases, 1991; 18:92-101 |
| 4. | J.N. Wasserheit. Epidemiological Synergy: Interrelationships between human immunodeficiency virus infection and other sexually transmitted diseases, Sexually Transmitted Diseases 1992; 19:61-77 |
| 5. | Johnson Phillip C.Testing for Syphilis, Dermatologic Clinic 1994; 12 Jan: 9-17 |
ICT INTERNATIONAL 271 De Goede Hoop Estate Noordhoek Cape Town South Africa Email :russell@ictdiagnostics.co.za Tel : + 27 82 441 1922 www.ictdiagnostics.co.za |  VNumber: 146234200 Effective date: 2020-04-13 |
Rev : 17 March 2022 |
|